|
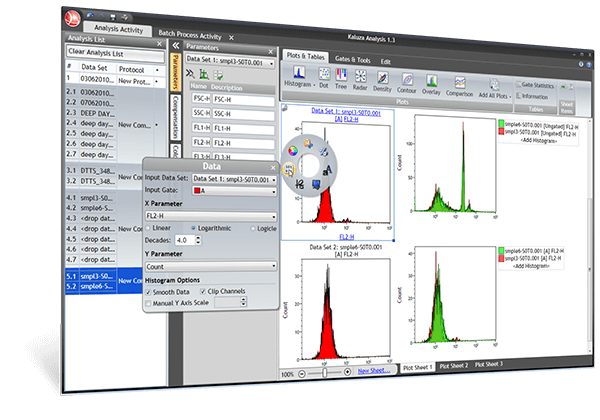
Plot Types
|
Add All Plots, Cell Cycle Analysis Plots, Comparison Plots, Contour Plots, Density Plots, Dot Plots, Histograms, Overlay Plots, Radar Plots, Tree Plots |
|
Gates
|
Auto Gates, Boolean Gates, Ellipse Gates, Hinged Quadrant Gates, Linear and Divider Gates, Polygon Gates, Quadrant / Staggered Quadrant Gates, Rectangle Gates, Showing Gated Populations as Rare Events, Viewing Gate Logic |
|
Statistics
|
% Gated, % Grandparent, % Total, Arithmetic Mean, Coefficient of Variation (CV), Half Peak of Coefficient of Variation (HPCV), Median, Mode, Number, Standard Deviation (SD) |
|
Compensation
|
Accounting for Auto-fluorescence, Adjusting Compensation, Automatic Compensation Calculation, Importing a Compensation File, Resetting Spillover and Auto-fluorescence Vector Values, Saving a Compensation File |
|
Reporting
|
Approval Signature, Comparing Report Sheets, Configure Report Sheets including layout formatting, Export information to Laboratory Information System, Export plots and data for use in other applications, Master Page, QC Reports including Levey-Jennings |
|
License Type
|
Individual User |
|
Number of Users
|
1 |
|
License Term
|
1 Year |